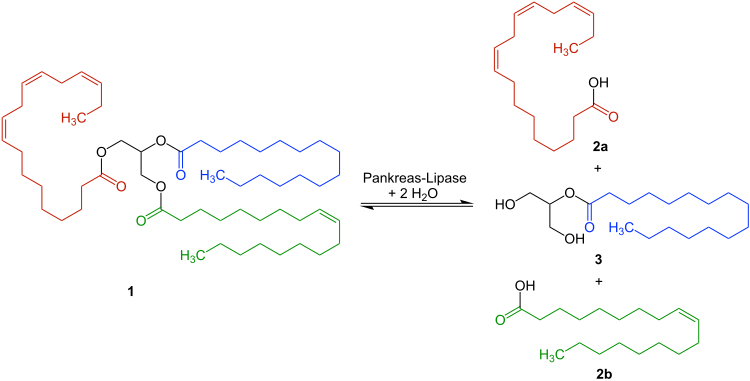
Studies on the Substrate and Stereo/Regioselectivity of Adipose Triglyceride Lipase, Hormone-sensitive Lipase, and Diacylglycerol-O-acyltransferases* - Journal of Biological Chemistry

Controlling the enantioselectivity of enzymes by directed evolution: Practical and theoretical ramifications | PNAS
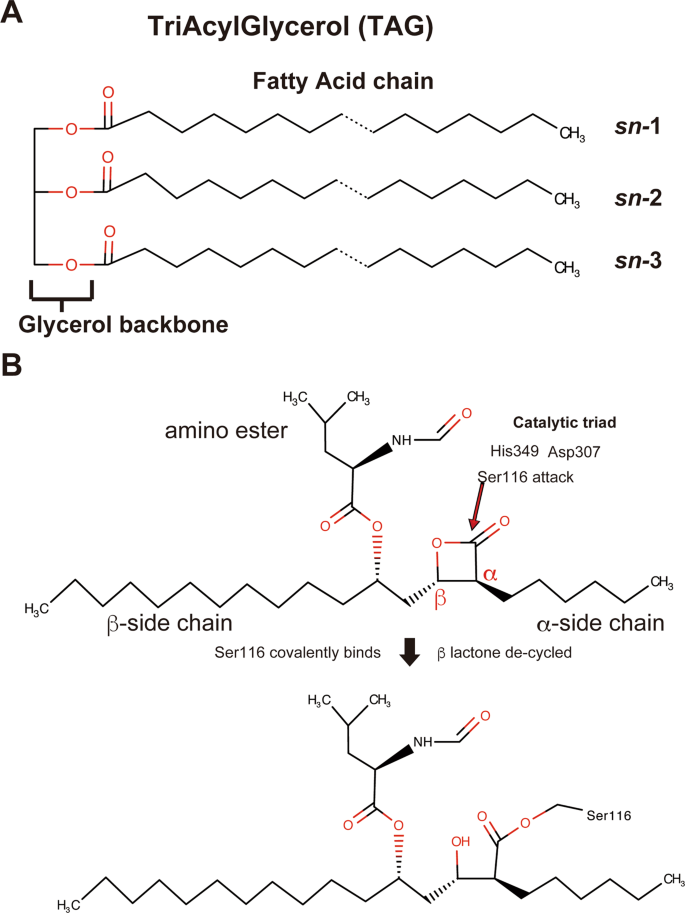
Crystal structure of pathogenic Staphylococcus aureus lipase complex with the anti-obesity drug orlistat | Scientific Reports
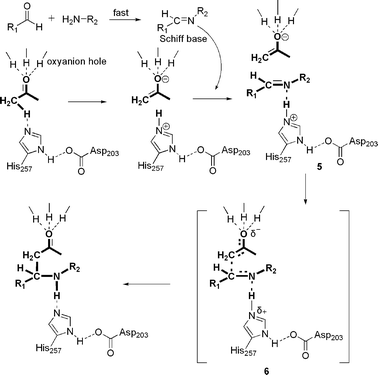
Lipase-catalysed direct Mannich reaction in water : utilization of biocatalytic promiscuity for C–C bond formation in a “one-pot” synthesis - Green Chemistry (RSC Publishing) DOI:10.1039/B817524A

SciELO - Brasil - Aspects on the catalysis of lipase from porcine pancreas (type VI-s) in aqueous media: development of ion-pairs Aspects on the catalysis of lipase from porcine pancreas (type VI-s)
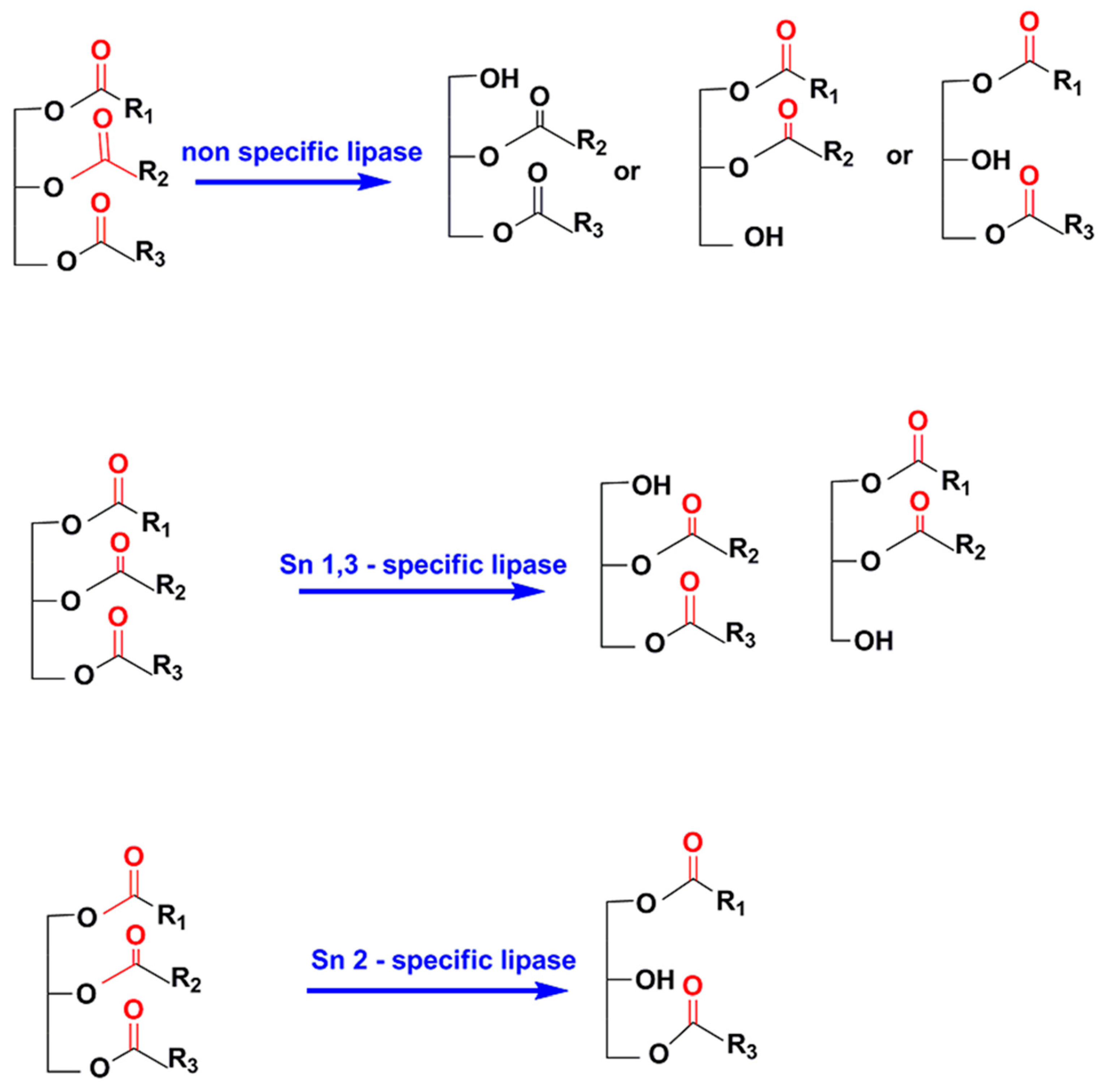
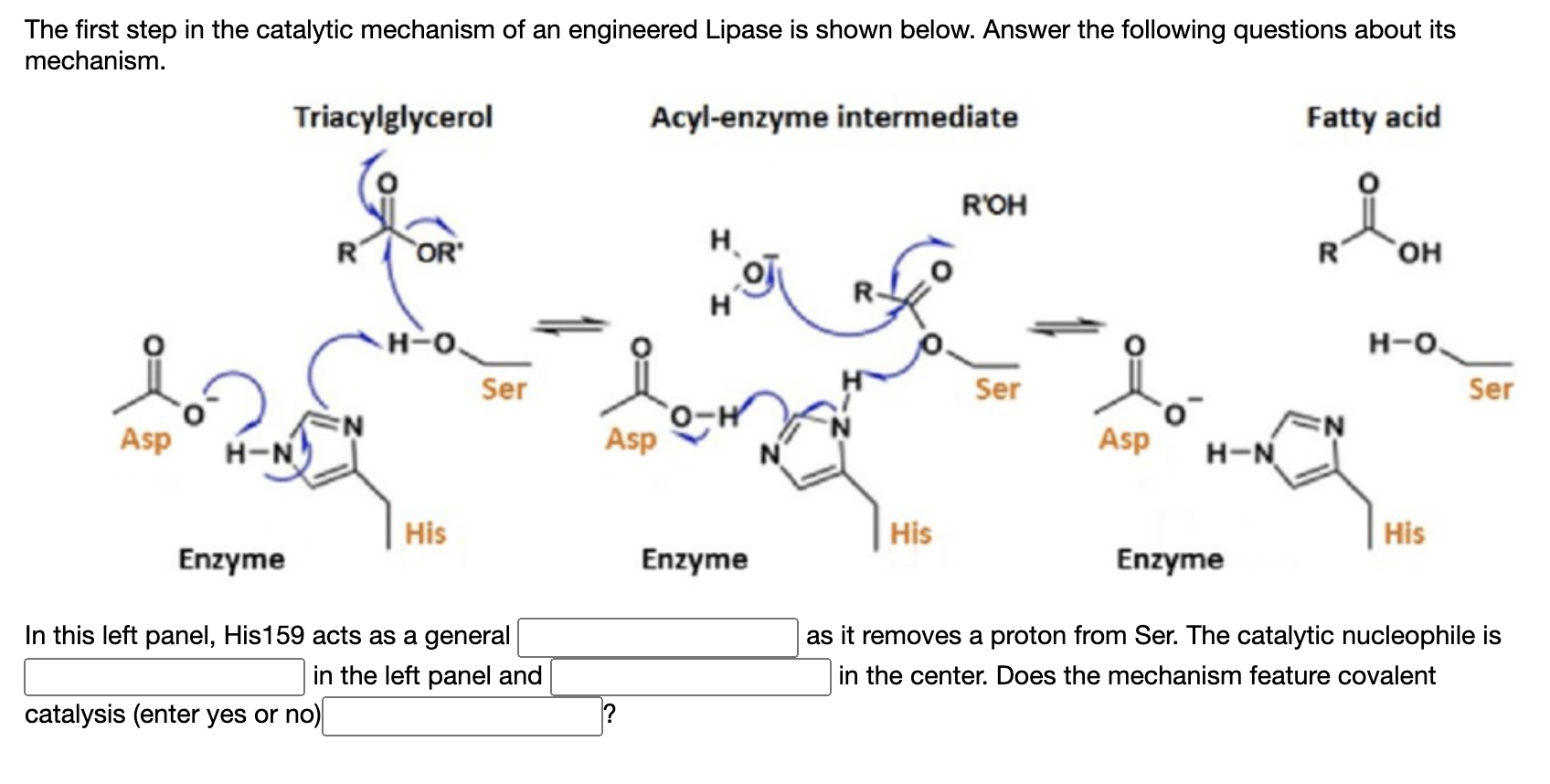
Catalysts | Free Full-Text | Main Structural Targets for Engineering Lipase Substrate Specificity | HTML
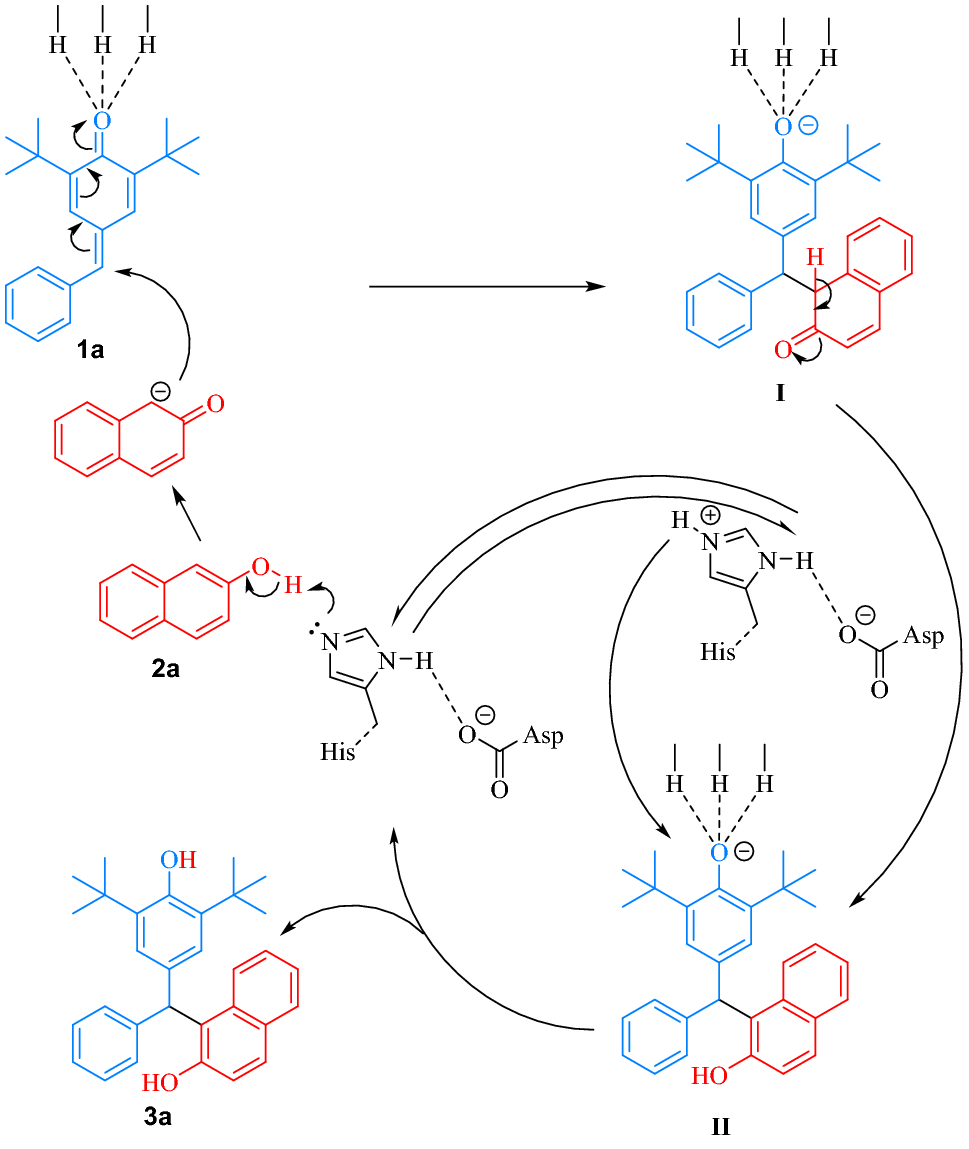
Figure 5 | Lipase-Catalyzed Highly Efficient 1,6-Conjugated Addition for Synthesis of Triarylmethanes | SpringerLink

SciELO - Brasil - NANOBIOCATALYTIC SYSTEMS BASED ON LIPASE-Fe<sub>3</sub>O<sub>4</sub> AND CONVENTIONAL SYSTEMS FOR ISONIAZID SYNTHESIS: A COMPARATIVE STUDY NANOBIOCATALYTIC SYSTEMS BASED ON LIPASE-Fe<sub>3</sub>O<sub>4</sub> AND CONVENTIONAL SYSTEMS ...
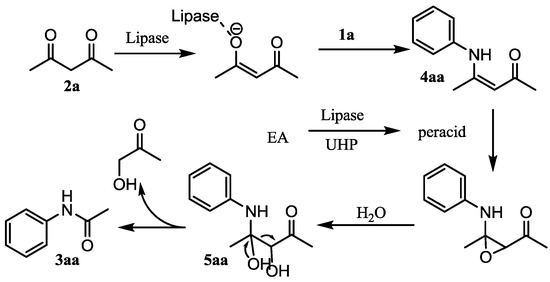
Catalysts | Free Full-Text | Lipase-Mediated Amidation of Anilines with 1,3-Diketones via C–C Bond Cleavage | HTML
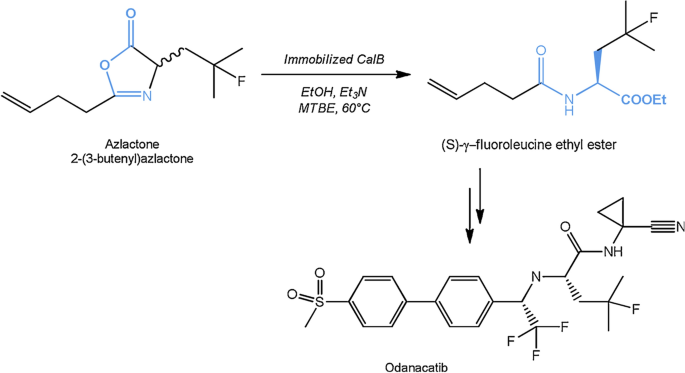
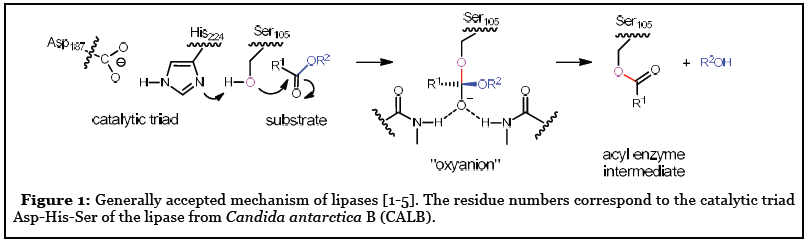
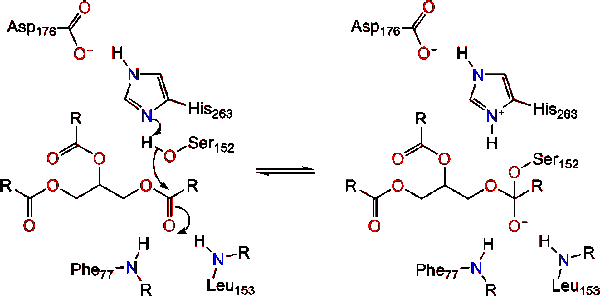
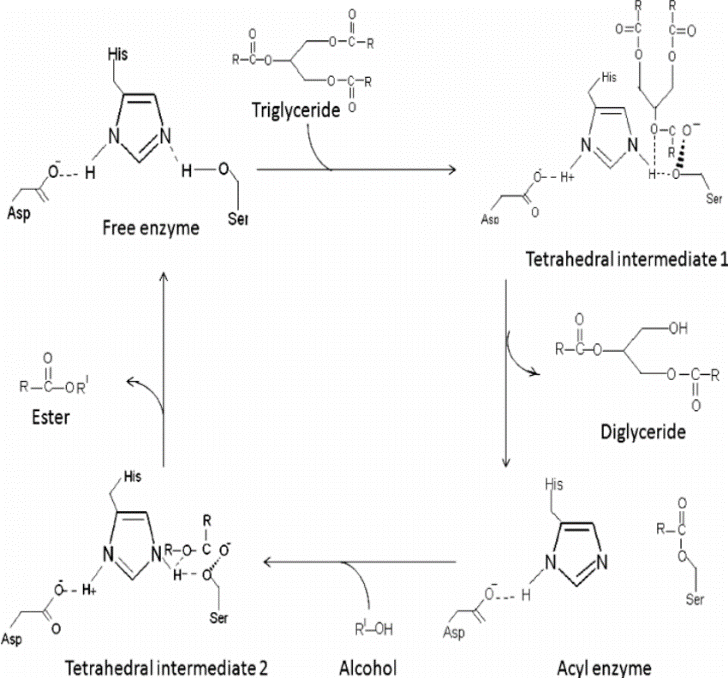
![Catalytic mechanism of lipases, adapted from [2]. | Download Scientific Diagram Catalytic mechanism of lipases, adapted from [2]. | Download Scientific Diagram](https://www.researchgate.net/profile/Georgina-Sandoval/publication/300081466/figure/fig5/AS:668455300763651@1536383571214/Catalytic-mechanism-of-lipases-adapted-from-2.png)




.png)